Biotech / Pharmaceutical
Cell growth is a complex process. Pharmaceutical manufacturing, which relies on the production of live cells, requires balancing the amounts of intake nutrients and accounting for the metabolic processes and metabolites produced during growth. Very often, cell-based production is a very high-cost manufacturing process so waste avoidance and process optimization are crucial.
The use of spectroscopic sensors for bioprocess monitoring is a powerful tool as a mechanism to design, analyze and control the pharmaceutical manufacturing process through the measurement of critical process parameters which will affect critical quality attributes. In-line measurements are particularly important during the cost-intensive manufacturing of biopharmaceuticals in order to facilitate any early process fault detection. This will help to minimize the risk of contamination and observe real time product release.
Featured Applications
CHO Cell Cultivation
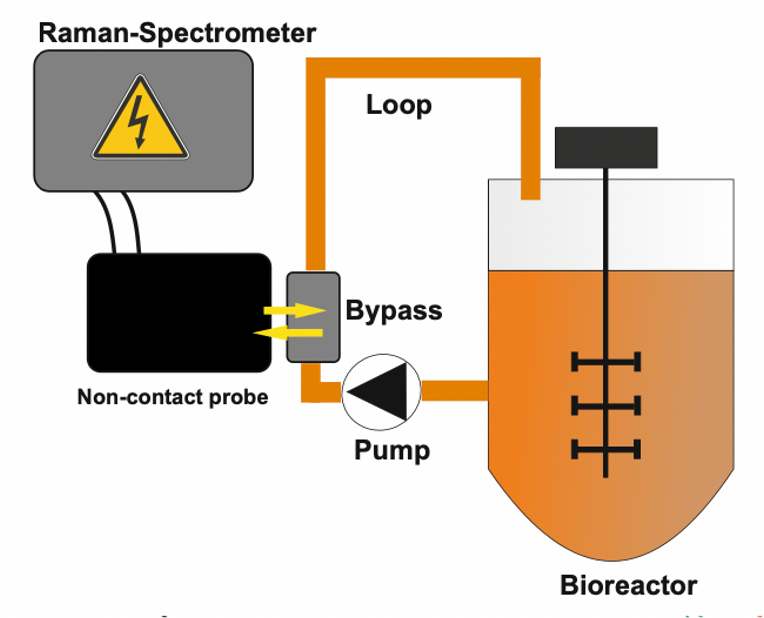
Bioprocess monitoring of CHO cell cultivations.
Our non-contact Raman Process Spectrometer System is used for monitoring of CHO cell cultivations for monoclonal antibody production in a 2L stirred tank bioreactor.
- Raman spectra obtain chemical information about the bioprocess.
- Spectral trajectory can be measured through the glass.
- Baseline shift comprises information about the cell concentration.
- No reference data needed – only spectral data.
- Detect deviations from standard batches.
Monitoring Monoclonal Antibody Breakthrough Curves On Chromatographic Columns

One of the biggest challenges in the preparation of monoclonal antibodies is often the purification process using chromatography on an industrial scale. This chromatographic separation processes need to be well-controlled to obtain the maximum product yields with minimal contamination.
Our high performance Raman spectrometer can monitor the breakthrough curves in-situ indicating when a column is completely saturated which is crucial for the optimization of chromatographic antibody purification.
Process Monitoring for Wet Granulation

Granulation processes are critical in the pharmaceutical manufacturing as this is where the concentration of the active pharmaceutical ingredient (API) in the final tablet or capsule can be controlled.
This wet granulation process must be carefully controlled for optimal product manufacturing. Care must be taken to avoid segregation of the components of the powder mix to provide a good, homogenous mixture via the formulation of granules.
With our proven Near-Infrared (NIR) spectrometer, it can provide a non-invasive technique to monitor product homogeneity and particle size distributions. By monitoring the spectral signatures associated with particle size distribution, moisture content and density, it was possible to characterize the formulation and better understand the underlying granulation process.
Biocatalytic Synthesis

Process monitoring of biocatalytic synthesis of a non-steroidal anti-inflammatory drug. The reaction of interest is the biocatalyzed synthesis of the drug.
With our Raman spectrometer immersed in the reaction mixture, the spectrometer is an excellent option for inline monitoring of biocatalytic synthesis in aqueous solution without spectral disturbance caused by water.
Glove Box

A glove box in a laboratory is a system that establishes a controlled environment, separate from the surrounding atmosphere in which materials can be manipulated. Maintaining an atmosphere that is almost void of oxygen (O2) is critical when storing and manipulating reactive substances within a glovebox.
Our specialized Oxygen (O2) analyzers are ideal for detecting ppm or “trace” level of oxygen within these enclosed atmosphere.
Oxygen Deficiency Monitoring

Regardless of the application of the compressed gas in the laboratory, if the gas is stored in any volume, then any leak in the storage or distribution systems or an unplanned release of the inert gas will quickly displace the normal atmospheric oxygen within the lab. Oxygen concentration can rapidly drop to dangerous level resulting in asphyxiation or death of lab personnel.
Our Oxygen Deficiency Monitor will detect low oxygen levels in the atmosphere and sound an alarm to alert individuals in the area to the danger.
Automated Control of Glucose Concentration in CHO cell Cultures

The active ingredients of biopharmaceuticals often have complex and unstable molecular structures as compared with chemical synthetic drugs. Therefore, quality control is particularly important. An automated real-time monitoring and advanced control of the cell culture is required to efficiently drive the cultivation process of biopharmaceutical production.
Equipped with an FT-NIR technique and a mathematical model software, our advanced, integrated control system can sense and predicts the future state of glucose consumption and lactate generation of the CHO cell culture. This helps to provide real-time monitoring of glucose, lactate, and live cell density with high accuracy throughout the entire culture period.